Researching Treatments for MPS-1 Hurler
Precision Delivery For Gene/RNA Therapeutics
Hurler Syndrome, A Focused Lead Programme With Platform Potential
Partnering To Advance RNA Therapeutics
Support MPS I (Hurler Syndrome) Research
A preclinical biotechnology company advancing tissue-targeted delivery platforms for genetic medicines. Our lead programme, DAWN-01, is an investigational in vivo stealth lentiviral vector for MPS-1 Hurler, supported by preclinical proof-of-concept data. In parallel, we are advancing targeted delivery platforms for CNS and musculoskeletal applications.
A dual-platform strategy, lead programme progression toward IND/CTA readiness, and platform expansion across high-need indications.
42% Average Reduction in Toxic GAGs
Reduction in Toxic GAG Accumulation (preclinical)
Transforming Lives with Gene Therapy for Hurler Syndrome
Our patented sLV platform delivers breakthrough treatment for MPS-1 Hurler patients with unmet medical needs
- 42% average reduction in toxic GAG accumulation achieved in preclinical studies
- First therapy to address residual disease after bone marrow transplantation
- Patented sLV technology targeting bones, joints, and cartilage
- Accelerated FDA approval pathway for a rare disease
- Potential to improve both lifespan and quality of life for affected patients
- Preclinical proof of concept achieved in MPS-1 Hurler models, including biomarker improvement (GAG-related endpoints).
- Designed for a high unmet-need population, including residual disease after HSCT.
- 24-month plan to IND/CTA readiness, subject to funding and stage-gate outcomes.
sLV Platform
Patented stealth lentiviral vector (sLV) platform engineered for targeted delivery to bones, joints and cartilage — addressing residual disease after bone marrow transplant.
Indication: MPS-1H (Hurler Syndrome)
Development status: Preclinical — demonstrated 42% average reduction in toxic GAG accumulation in target tissues.
DAWN-01 is an investigational in vivo stealth lentiviral vector (sLV) designed to deliver a functional IDUA gene for MPS-1 Hurler, with a focus on addressing residual disease that persists after standard care. In preclinical MPS-1 models, Dawn’s data show meaningful biomarker improvement, including reduced GAG burden and normalisation of urinary GAG readouts.
14× Brain to Blood Ratio Efficiency
Peer-reviewed Research
Angew. Chem. Int. Ed. 2025 | doi.org/10.1002/anie.202500247
Revolutionary Brain Delivery: 14x More Effective Than Current Systems
Our patented iLNP technology crosses the blood-brain barrier to treat neurological diseases
- 14-fold superior brain-to-plasma ratio compared to existing delivery systems
- Patented LNP formulation with transferrin receptor targeting
- Successfully penetrates the blood-brain barrier and reaches neurons
- Validated delivery to deep brain regions: thalamus, hippocampus, cerebellum
- Platform for treating Alzheimer's, autism, and other CNS disorders
iLNP Platform
Patented lipid nanoparticle (iLNP) formulation engineered for transferrin receptor targeting and validated deep brain delivery.
Key metric:
- 14× improved brain-to-plasma delivery vs current systems.
- Joint-targeting iLNP data showed 7-fold GFP expression increase and 5-fold NOTCH1 knockdown in vivo.
Up to 6.5 brain-to-serum ratio: Published TfR aptamer conjugate study supports BBB-targeting strategy for nucleic acid delivery.
Applications: Alzheimer’s disease, autism spectrum disorders, other CNS indications.
Our Mission Statement:
At Dawn Therapeutics, our mission is to democratise RNA therapy by making transformative treatments more accessible and affordable for patients. We are building next-generation delivery and manufacturing technologies designed to reduce cost, improve tissue targeting, and overcome key barriers that limit current therapeutic approaches. Our lead focus includes MPS-1 Hurler, while our platform technologies are designed to extend across multiple serious diseases.
Targeted Lipid Nanoparticle (iLNP) Platform Brain
A tissue-targeted LNP platform designed to carry DNA, RNA, and gene-editing payloads with high precision using proprietary targeting ligands, including transferrin receptor targeting for CNS delivery and antibody-enabled targeting for joint/cartilage delivery.
1. Internal Dawn preclinical data show 7-fold GFP expression increase in joints and 5-fold NOTCH1 knockdown in joint tissue in vivo.
2. Published TfR aptamer conjugate work supports the CNS targeting strategy, with brain-to-serum ratios up to 6.5 and neuronal localisation in mice.
3. Manufacturing strategy is positioned for scalability and lower cost, with an illustrative variable COGS target iLNP products at scale assumptions.
Stealth Lentiviral Vector (sLV) Platform Hurler Syndrome
A third-generation in vivo lentiviral platform engineered for safer and more targeted gene delivery. The platform incorporates immune-evasion features, APC detargeting target sites, tissue-restricted expression (including liver-focused promoter strategies), and a tEGFR safety switch. It underpins DAWN-01 for Hurler syndrome.
1. Preclinical biomarker improvement in MPS-1 Hurler models, including urinary GAG normalisation and tissue-level GAG reductions.
2. Built-in safety and translational risk-mitigation features support regulatory readiness planning.
Patients & Families
We work hand in hand with patients, families, and advocacy groups to provide clear information, compassionate support, and access to cutting-edge therapies that can transform lives.
Science & Innovation
Our proprietary delivery platform enables RNA therapies to reach precise target tissues, opening new possibilities for treating rare and complex diseases at their root cause.
Partnering for Impact
From early research collaborations to licensing agreements, we work with innovators worldwide to accelerate breakthroughs and bring them to patients faster.
Disease Area
MPS-1 Hurler Syndrome (MPS-1H)
A rare genetic disorder caused by enzyme deficiency, leading to progressive tissue and organ damage.
DAWN-01: MPS-1 Hurler Syndrome (MPS-1H)
Overview
DAWN-01 is an investigational, in vivo "stealth" lentiviral vector (sLV) designed to deliver a functional IDUA gene to address Hurler syndrome (MPS-I H), a severe and life-limiting lysosomal-storage disorder.
Current Status
We have successfully achieved the pre-clinical in vivo proof of concept. Our next objective is to raise funds via investment or donation to perform GMP, Toxicology, and Phase 1 Clinical Trial.
Our aim is to progress DTX-101 to first-in-human clinical testing following successful completion of these studies and regulatory review.
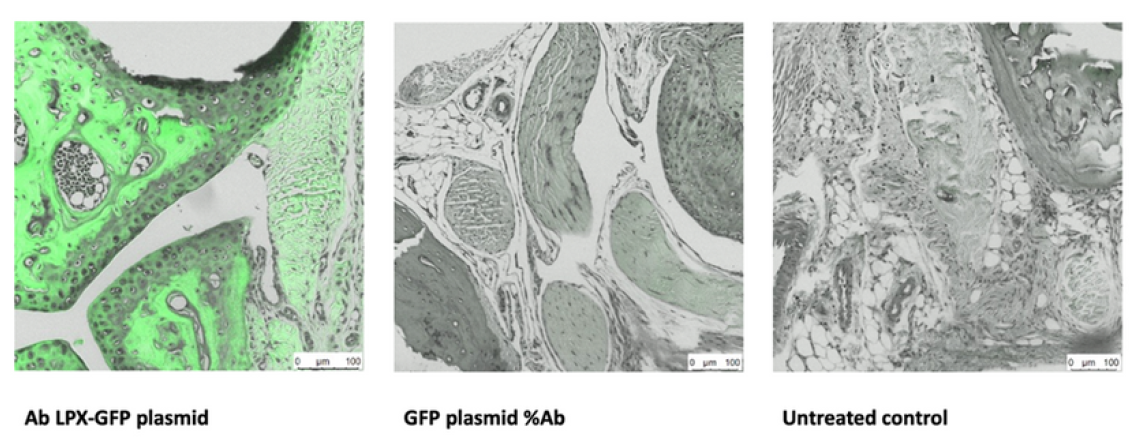
Histology: Joint sections following targeted delivery
Left: Ab LPX-GFP plasmid. Middle: GFP plasmid %Ab. Right: Untreated control. Bright green signal in treated joints confirms expression of GFP within cartilage and joint structures. Scale bars 100 µm. Leica LMS, GFP and light channels.
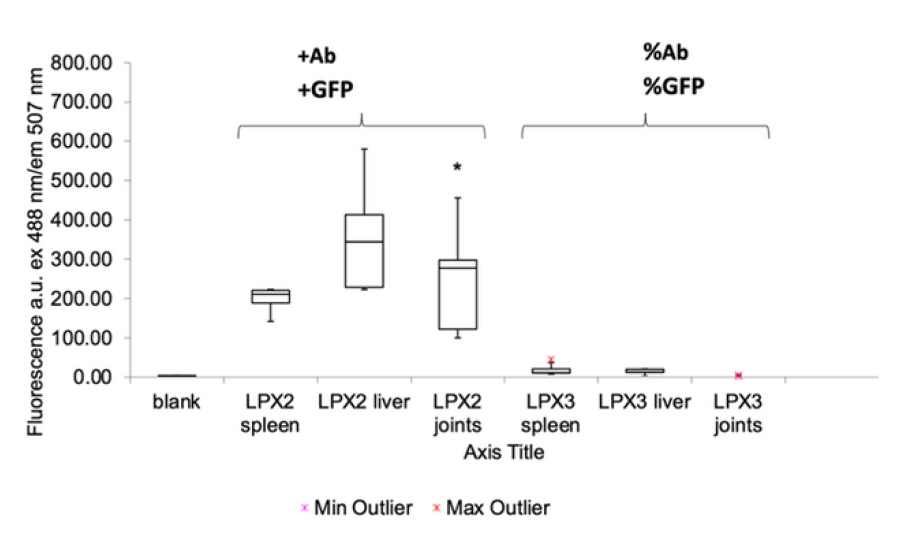
Results: GFP expression increased seven fold in joints in vivo
Box plots of GFP fluorescence (ex 488 nm, em 507 nm) demonstrate higher reporter expression in LPX2 joints versus controls. ANOVA p = 0.023 at 95 percent significance. N = 5 per group. Min and max outliers indicated.
Conclusion:
Histology shows clear signal localised to joint cartilage in treated animals, with minimal background in controls. Box plot confirms about seven-fold higher reporter expression in joints. Effect is statistically significant (ANOVA p = 0.023, N = 5 per group). Together, these data validate effective, targeted in vivo delivery & support advancement to first-in-human studies.
High-yield summary
In vivo cartilage and joint targeting was validated by histology and quantitative fluorescence readouts, supporting further development of DAWN-01.
- 1Seven fold higher reporter expression in joints compared with controls. Current delivery systems achieve a brain-to-blood ratio of less than or equal to 0.5, so our delivery is 14 times more efficient. In simple terms, more of the medicine reaches the target tissue and less is wasted in the bloodstream, which can mean lower doses and fewer side effects.
- 2Robust tissue localisation in H and E sections with bright signal in treated joints. This means standard microscope stains show the signal sitting inside the joint structures where it is needed, not spread randomly, which is what you want for precise treatment.
- 3Consistent performance across animals with statistical significance at 95 percent. In practice, the results are unlikely to be due to chance and can be reproduced, which increases confidence that the effect is real.
- 4Translatable platform for IDUA and related payloads in musculoskeletal disease. The same delivery vehicle can carry the actual therapeutic gene or other cargos to cartilage and bone, helping shorten the path from lab findings to real treatments.
- 5Readouts align across modalities: qualitative histology and quantitative fluorescence. Both the pictures and the numbers tell the same story, which is a strong cross-check that the technology is doing what it should.
- 6Platform is payload-agnostic and designed to support IDUA delivery for MPS-I H. That means we can swap in different genetic instructions when needed, including IDUA for Hurler syndrome, without redesigning the whole system.
These findings provide the scientific basis to progress to GMP manufacture, formal toxicology, and first-in-human evaluation.


At Dawn Therapeutics, we are advancing a new generation of targeted delivery systems for genetic medicines, combining stealth lentiviral vector engineering and tissue-targeted lipid nanoparticle technologies. Our platforms are designed to improve delivery precision across high-need tissues, including the brain, joints, cartilage, and bone, with a focus on scalability and translational readiness in preclinical development.
Precision Targeting
Advanced vector engineering delivers therapies only where they are needed, reducing off-target effects.
Payload Versatility
Supports DNA, RNA, and gene-editing payloads, including mRNA, siRNA, and other programmable nucleic acid formats.
Clinical Readiness
Built with scalable manufacturing and GMP-aligned development planning to support efficient translation from preclinical studies toward the clinic.
Programme Pipeline
DAWN-01
sLV / CNS
Hurler Syndrome (MPS I)
Preclinical: Awaiting Funds
Work With Us to Advance Breakthrough Therapies
Join our team or collaborate with us to turn cutting-edge science into real-world solutions for patients worldwide
